Discovery Spotlight
Huy Dinh, PhD
Assistant Professor
Department Oncology
Immune heterogeneity and plasticity during cancer progression
We utilize systems and computational biology approaches that leverage high-dimensional data from multi-omics genome-wide (genomics and epigenomics) and single-cell assays. We develop bioinformatics methods and pipelines to answer the following questions.
1) How does the tumor immune microenvironment (TIME) influence immunotherapy? We focus on Immune checkpoint blockade (ICB), which operates by blocking cellular communication (e.g. ligand-receptor interactions) that impacts the tumor-killing ability of T-cells. Emerging evidence has implicated myeloid cells (with pro- and anti-tumoral functions) in aiding ICB efficiency. We aim to identify mechanisms of crosstalk between specific myeloid and T-cell subsets that are involved in orchestrating immune responses before and after ICB therapy.
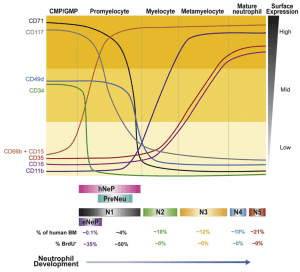
2) What is the role of neutrophils in cancer? Neutrophils, the most abundant blood cell-type, are elevated in peripheral blood of cancer patients and associated with poor prognosis for multiple types of cancer. However, neutrophil heterogeneity and plasticity in the tumor microenvironment are not incompletely understood. We discovered an early unipotent neutrophil progenitor in human bone marrow marked with a potential role in cancer. We are analyzing the types of neutrophils that are derived from the neutrophil precursors and how they diversify in circulation, at tumor and metastasis sites. We are testing if neutrophil subpopulations and their markers can predict cancer progression and response to treatments.
3) Can cancer treatment outcomes be predicted based on blood-based biomarkers? Identification of predictive markers, especially by non-invasive approaches, is urgently needed. We are defining specific immune signatures, with a focus on myeloid cells, from the blood of cancer patients that can predict cancer treatment outcomes.
Publications:
https://www.ncbi.nlm.nih.gov/myncbi/1Deyjxj9VFe5v/bibliography/public/